Atrial Fibrillation & Therapeutic Innovation
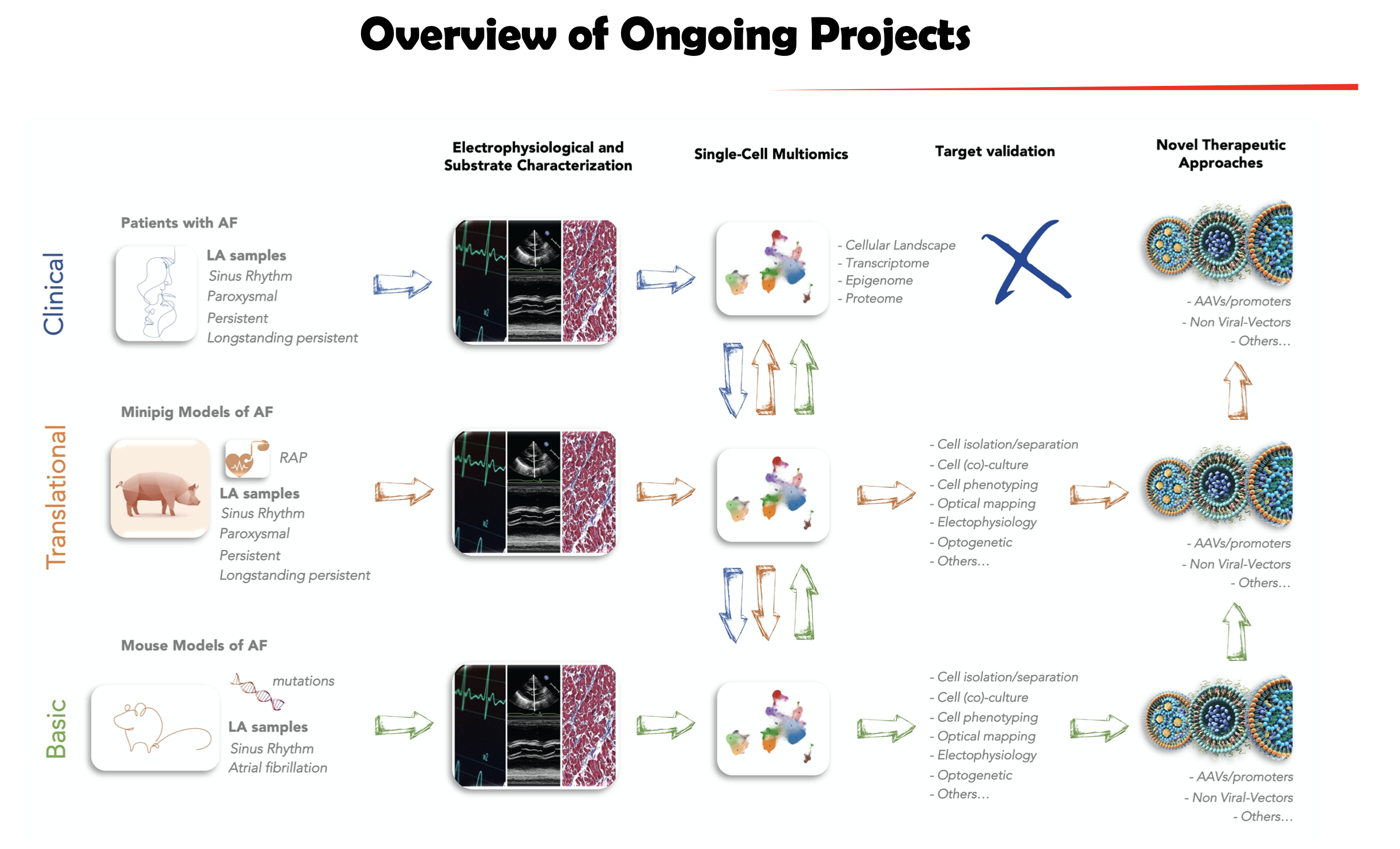
As the most common cardiac arrhythmia worldwide, atrial fibrillation (AF) is a growing epidemic with a prevalence estimated to rise to 12.1 million in 2030. It presents a serious health burden due to high morbidity and mortality rate. Patients with AF have an increased risk of stroke and heart failure. AF is defined as paroxysmal (≤ 7 days), persistent (> 7 days) or long-standing persistent (≥ 1 year) depending on the duration of the arrhythmia. Beside pharmacological therapy and electrical cardioversion which have limited efficacy, patients can also be treated with catheter ablation. However, while patients with paroxysmal AF have the highest success rate, ablation is generally less effective in non-paroxysmal AF. Despite the extensive research efforts made in the past decades, the determinant trigger for the progression of paroxysmal to persistent AF remains poorly understood. In this context, a better mechanistic understanding of the cellular and molecular process forming the substrate of AF is expected to foster the development of safer and more effective treatment approaches.
With translational approaches and in close collaboration with clinicians, we aim to decipher the mechanisms underlying AF and its progression. To this end, we combine high-throughput molecular biology techniques such as transcriptomic, proteomic and lipidomic, with animal models (from transgenic mice to AF model of higher species) and state-of-the-art electrophysiological studies (in vitro and in vivo). The targets identified in this context will allow the development of innovative approaches for the treatment of AF by means of specific vectors (e.g. viruses, molecular compounds) targeting a defined cardiac cell population (precision medicine). Our ultimate goal is to offer new therapeutic options in terms of personalized medicine for the patient suffering from AF.
Contact
PD Dr. Ange Maguy
Head of the Atrial Fibrillation & Therapeutic Innovation Lab
E-mail: ange.maguy@unibe.ch